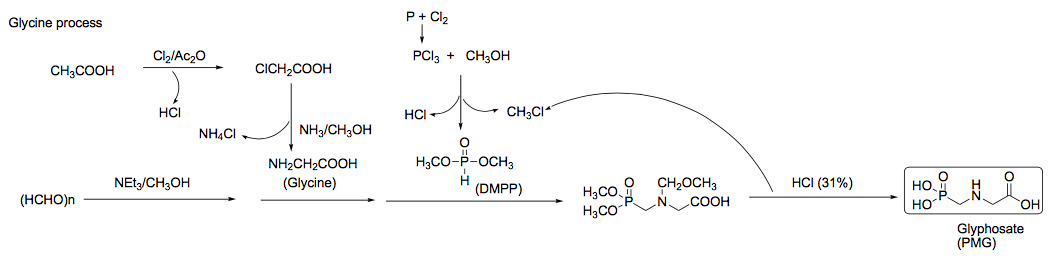
The third way is called the Glycine Process.
This process, which is the most widely used process, begins with paraformaldehyde ((HCHO)n) undergoing depolymerization, which is the process of a polymer (many molecules all strung together to form a long chain or a complex structure) turning in to one or many monomer(s) (a molecule that has potential to become a polymer). This depolymerization of paraformaldehyde is catalyzed by triethylamine (NEt3) in anhydrous methanol (CH3OH). It reacts with glycine (NH2CH2COOH) and dimethylphosphonate (DMPP), to which concentrated hydrochloric acid is added and the whole thing is heated to produce glyphosate.
The glycine used in the reaction is prepared by first reacting acetic acid (CH3COOH) with chlorine to produce chloroacetic acid (ClCH2COOH). This chloroacetic acid is then reacted with ammonia (NH3) in anhydrous methanol (CH3OH) to produce glycine.
Dimethylphosphonate (DMPP) is prepared by reacting phosphorous chloride (PCl3) with anhydrous methanol. The methyl chloride (CH3Cl) byproduct is reused as a start material for creating silicon products.
The glycine used in the reaction is prepared by first reacting acetic acid (CH3COOH) with chlorine to produce chloroacetic acid (ClCH2COOH). This chloroacetic acid is then reacted with ammonia (NH3) in anhydrous methanol (CH3OH) to produce glycine.
Dimethylphosphonate (DMPP) is prepared by reacting phosphorous chloride (PCl3) with anhydrous methanol. The methyl chloride (CH3Cl) byproduct is reused as a start material for creating silicon products.